
“Estimating Surface Area of Silver Grains: From Measurements to Calculations“
Greetings, metallurgy enthusiasts! Today, we’re delving into the practical side of estimating the surface area of silver grains used as anodes in a silver-plating bath. Follow along for a detailed breakdown:
Step 1: Volume Measurement
In Photo #1, we poured 248 grams of silver grains into a container, measuring a volume of 34 cubic centimeters.
[Include Photo #1 – Silver grains in the container]

Step 2: Diameter Measurement
After measuring several silver grain diameters and averaging them to 3.9 millimeters (Include Photo #2- grain measurements)

Step 3: Calculating Surface Area of a Sphere
The formula for the surface area of a sphere is A=4πrr, where r=D/2. Plugging in the values, we find the surface area of one average silver grain: 47.75square millimeters or 0.477square centimeters.
Step 4: Weight Measurement
As seen in Photo #3, the weight of 25 grains of silver is 7.06 grams. We can say that on average, each grain weighs 0.28 grams (7.06/25).

Step 5: Total Surface Area Calculation
Now, let’s calculate the total surface area per 1000 grams of silver grains. Using the average weight, we find that 1000 grams of silver grains will have a volume of 1703.5 cubic centimeters.
(1000 grams of silver grains) ÷ (0.28 grams per grain )× (0.477 cm2)=1703.5 cm2
Step 6: Scaling for 1000 Cubic Centimeters
To calculate the surface area of 1000 cubic centimeters of silver grains, we scale up. Given that 1000 cubic centimeters of silver grains weigh 7294 grams:
(calculation is as follow 7294=1000/34*248) . we can now calculate 7294 gr generate 26050 cubic centimeters (calculation is as follow 7294/0.28=26050).
There you have it! By meticulously measuring, averaging, and calculating, we’ve successfully estimated the surface area of silver grains per 1000 grams weight and 1000 cc volume, shedding light on the intricacies of silver plating.
Notes:
- Each lot of grains should be measured in order to indicate above values
As size may change from lot to lot.
- Grains are ideally to generate big surface area but demand some more
Work and follow up.
- Surface of the anode is a key factor to the silver-plating control.
- This method is good for any metal grains

GemOro Auracle Pen Probe
GemOro Auracle Pen Probe | Plug & Play Universal Replacement for AGT1 AGT3 Digital Gold & Platinum Testers | Precision Tool with Expert Professional Reading

Triple Diversification: Expanding Your Investment Strategy Beyond Borders and Banking
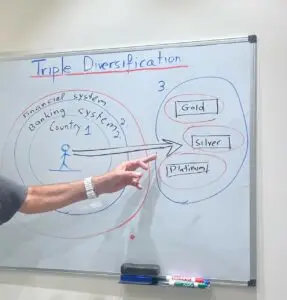
Triple Diversification: Expanding Your Investment Strategy Beyond Borders and Banking
Triple Diversification: Expanding Your Investment Strategy Beyond Borders and Banking In the realm of physical investments, diversifying one’s portfolio is often associated with the acquisition

Electrical Conductivity of Precious Metals
SummaryThis article explores the electrical conductivity of precious metals and their alloys, focusing on its measurement in Siemens per meter and variations across different materials.


